PVC COMPOUNDING:
POLYVINYL CHLORIDE:
PVC is a product based on two of the earth’s natural resources, salt and oil. Salt water electrolysis yields chlorine (in addition to caustic soda and hydrogen). Ethylene can be derived from naphtha when oil is refined. Chlorine and ethylene can be combined to form the monomer, vinyl chloride (VCM). PVC results from the polymerisation of vinyl chloride.

(PVC CHEMICAL CONFIGURATION)
PVC cannot be processed on its own due to its very low thermal stability and high melt viscosity. Therefore, it is necessary to combine with the polymer a number of suitable additives to give a wide and varied range of properties to satisfy many different end-use applications. It is this scope for generating a wide range of properties through appropriate formulation that gives PVC a broad application coverage that is unmatchedby any other commodity thermoplastic. Its durability (85% is used in medium- to long-term life spanapplications), linked to its cost effectiveness, has made it the second largest commodity plastic afterpolyethylene.
PVC COMPOUNDS:
PVC compounds are based on the combination of the polymer and additives that give the formulation
necessary for the end-use. The convention in recording the additive concentration is based on parts per hundred of the PVC resin (phr). The compound is generated by intimately mixing together the ingredients, which is subsequently converted into the gelled article under the influence of heat (and shear). Depending on the type of PVC and additives, the compound prior to gelation, can be a free-flowing powder (known as a dry blend) or a liquid in the form of a paste or solution.
PVC compounds can be formulated, using plasticisers, into flexible materials, usually called PVC-P.
Compounds without plasticiser for rigid applications are designated PVC-U.
HOMOPOLYMERS:
The VCM repeat unit is linked in a head-to-tail fashion and the number of repeat units can range between 500 and 1,500. This corresponds to a theoretical molecular weight range of approximately 30,000 to 95,000. The properties of PVC are influenced by its average molecular weight and its molecular weight distribution. The chemical structure deviates from the theoretical chain with chain branching occurring during polymerisation.
Commercial PVC polymers are essentially amorphous but also have a crystalline phase amounting to
approximately 10% and a two-dimensionally ordered hematic phase depending on processing history.
monomer droplets can agglomerate to a greater or lesser extent in the course of the polymerisation. The reaction can be chemically short-stopped when the desired conversion has been reached by the use of particular phenolics. The suspended droplets of liquid monomer are progressively converted to solid or porous PVC particles. Unreacted VCM is stripped from the polymer and water is separated by centrifuging. The polymer is then dried, typically in fluidised bed drying equipment. Prior to start-up of the next polymerisation cycle, the reactor is cleaned and coated with an antifouling agent.
Typical suspension polymerised PVC has a mean particle size of 100-150 µm with a range of 50-250 µm. The particles are complex and irregular in shape with a dense semipermeable skin.
S-PVC is the most common type of resin (around 85% of total PVC produced) used in both rigid and flexible applications and grades are formulated to meet an extensive range of requirements such as, high plasticiser absorption for flexible products, or high bulk density and good powder flow required for rigid extrusion.
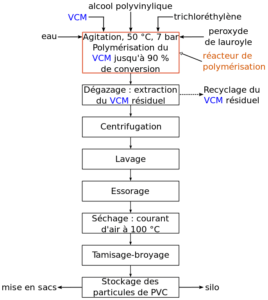
(S-PVC MANUFACTURING PROCESS)
Chlorinated PVC (CPVC)
CPVC is manufactured by chlorination of PVC polymer thereby raising the chlorine content from 56% to around 66%. Chlorination takes place primarily at the -CH2- groups along the polymer chain so that the resulting structure becomes virtually a copolymer of vinyl chloride with 1,2 dichloroethylene.
Chlorination of PVC reduces the forces of attraction between the molecular chains. CPVC is also essentially amorphous. Both of these factors allow CPVC to be stretched more easily and to a greater extent than PVC above its glass transition temperature. CPVC has a glass transition temperature approximately 50% higher than PVC but also has a higher melt viscosity in processing. The upper service temperature for CPVC is approximately 100 °C in comparison to PVC at 60 °C.
Pipe, mouldings and sheet are formulated for high temperature use based on CPVC or blends of
CPVC and PVC.
PVC Resin Characterisation:
Molecular Weight:
PVC resins are polymerised to different molecular weights to suit the particular processing technique and end-use application requirements. The resin contains a random distribution of molecular weights and number average, weight average and molecular weight distribution can be measured. Normally, however, for production control, the dilute solution viscosity of the resin is used and the molecular weight can be calculated from the Mark-Houwink equation expressed in terms of K value or viscosity number. Commercially available PVC grades are supplied in K values ranging from 57-80.
Particle Size
Various techniques are utilised to measure particle sizerange. Sieving is the most common technique,
particularly for suspension resins, and can be used tomeasure particles above 30 microns. For particle sizesbelow 100 microns, techniques such as sedimentation,optical and electrical sensing can be used.
Bulk Powder Properties
Bulk density and packed bulk (or tapped) density areimportant properties. The bulk density determines theweight of resin that can be stored in a vessel and theamount a mixer can hold. It also has a major influenceon extruder output rates. The bulk density of a resindepends upon its porosity, particle shape and particlesize distribution. For suspension resin, bulk density istypically in the range of 450-650 kgm-3.
The packed density is based on the ability of the resin to occupy the smallest space without physical distortion and is usually 10-30% greater than the bulk density. Another important consideration is powder flow time as it governs the speed and ease of polymer discharge from a silo or from a mixer. This is influenced by the PVC particle shape, size distribution and compound formulation ingredients.
Key Additives
PVC formulations can include many different types ofadditives which assist in imparting a large range ofphysical and chemical characteristics. This versatilityis the main reason why PVC has been so successful asa commodity thermoplastic, from medical applicationssuch as tubing and blood bags to long life applicationssuch as window frames and rainwater goods. Theunique polar characteristics of PVC permit a wide rangeof appropriate additives to be incorporated within thepolymer. The main groups of additives are:
Heat stabiliser, Plasticiser, Impact modifier, Process aid, Lubricant, Filler, Flame retardant/smoke Suppressant, Pigment, Blowing agent, Biocide, Viscosity modifier, Antistatic agent, UV absorber, Antifogging agent, Bonding Agent
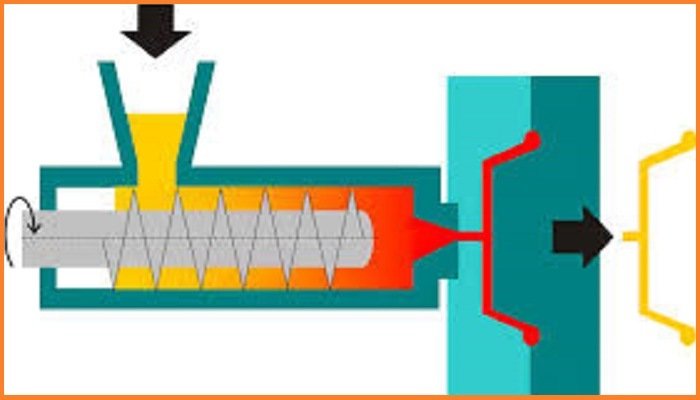
Processing Techniques
Prior to processing, it is necessary for the PVC and formulation additives to be combined intimately
together in some form of mixer. The main converting processes are:
• extrusion
• calendaring
• injection moulding
• extrusion/stretch blow moulding
• spreading/coating
• rotational moulding
• dip moulding
• slush moulding
PVC RESIN PRODUCERS IN GLOBAL MARKET:
- Shin-Etsu
- FPC-Formosa
- Geon
- Solvay
- EVC
- Kondea Vista
- Atofina
- Borden
- Norsk Hydro & many more..
Global Market Usage Analysis:
| Pipes/Fittings | 36% |
| Profiles | 13% |
| Rigid Film/Sheet | 7% |
| Flexible film/Sheet | 7% |
| Wire & Cable | 7% |
| Flexible Profile/Tubes | 5% |
| Flooring | 3% |
| Coatings | 2% |
| Bottles | 3% |
| Others | 13% |
PLASTISIZERS:
PHATHALATES;
regarding the possible negative impact of phthalates on the environment and human health due to alleged carcinogenic, endocrine modulating and reproductive effects. In particular, their use in toys, healthcare products and flooring has raised much debate and analysis at various levels concerning migration, blood compatibility.
The main phthalates under investigation are butyl benzyl phthalate (BBP), dibutyl phthalate (DBP),
di-2-ethyl hexyl phthalate (DEHP), diisononyl phthalate (DINP) and diisodecyl phthalate (DIDP).
These investigations include EU risk assessments based on sound science but the political positions on flexible PVC and phthalates cannot be ignored.
Adipates
PVC food packaging film (cling film) has been usedfor a number of years for wrapping fresh meat; for foodstorage; and for protecting food in the home.The most common plasticiser used in this application isdi-2-ethylhexyl adipate (DEHA), in combination withpolymeric plasticisers and epoxidised soya bean oil.DEHA is used in preference to phthalates due toimproved flexibility at lower temperatures and alsogives the necessary permeability to oxygen and watervapour to preserve the freshness of the food.
Heat Stabilisers
Lead Based Stabilisers
Primarily used extensively for PVC-U applications inEurope and the Far East viz. pipe, fittings and profile,lead stabilisers have been very cost effective heatstabilisers. For similar reasons, plus good insulationresistance, lead stabilisers have also been used in PVC-Pin wire and cable across the world. Their toxicityhazards have been well known and controlled,particularly in relation to worker exposure.
Bisphenol A/Alkylphenols
Bisphenol A has been used as an inhibitor at the PVCpolymerisation stage and as an antioxidant. There hasbeen some concern regarding possible health hazardsand its use has been voluntarily stopped or restrictedto levels that do not exceed the predicted no effectconcentration in waste water etc. The content of Bisphenol A in, and its migration from PVC film, has been determined by high performance liquid chromatography (HPLC).
Epoxidised Soya Bean Oil (ESBO)
ESBO is a heat stabiliser and secondary plasticiser usedin PVC-P applications. It has food contact approval upto certain limits but there have been someenvironmental/political issues concerning the possibleuse of genetically modified soya bean oil in themanufacture of ESBO.The migration of ESBO from PVC-P used in gasketsfor sealing baby food jars has been examined.
Solid Stabilisers
Lead Composition:
Lead compounds, usually based on a mixture of leadsalts, e.g., tribasic lead sulfate or dibasic leadphosphite,and lead soaps, e.g., dibasic or normal lead stearates,have been very cost effective heat stabilisers for manyyears, and function as HCl absorbers. They are particularly suitable for wire and cableelectrical insulation because of their complete nonconductivity and the inert nature of the chloridesformed with HCl.
Technical limitations to their use have been opacityand sulfur staining. Their cumulative toxicity has beenmainly a concern for worker exposure. Apart from wireand cable, their use has been restricted to PVC-Uapplications in Europe and Asia.
Non/low dusting products, which include the leadcomponents in a safe handling state, in combination
with lubricants (one-pack), have been available formany years in different product forms – flake, granule,tablet and dosage packs. These one-packs are tailoredto suit the processing technique, primarily extrusionand injection moulding, and end-use specification.
Calcium Zinc (Ca Zn) Based:
One of the most successful developments in recentyears has been the progress made in solid Ca Zn
stabiliser formulations to replace lead on a cost effectivebasis.Ca Zn formulations are a complex blend of calciumand zinc soaps together with acid acceptors and organicco-stabilisers. Efforts have been made to increase theacid absorption capability in these systems.
The role of the co-stabiliser is also critical inmaintaining initial colour and early colour hold on
processing. Alternativecalcium carboxylates have also been evaluated.Various Ca Zn systems are now available for pipe. fittings, profile and wire and cable, from suppliers such as Baerlocher and Chemson.Solid Ca Zn systems have also been developed for PVC-Papplications as replacements for liquid Ba Zn stabiliserswhere volatility and emissions may be an issue.Non dusting product forms have also been developed, due to the light and fluffy nature of the Ca Znsoaps (stearates).
Liquid Stabilisers
Organotin Compounds:
Organotin compounds are primarily based on alkyl tinsuch as methyl, butyl or octyl derivatives, usuallymixtures of di-alkyl and mono-alkyl. They can be furtherclassified as mercaptides or carboxylates based ondifferent carboxylic acids. The main features of tinstabilisers are good initial and long-term colour holdcoupled with excellent clarity, linked to their goodcompatibility. The most powerful compounds arethe thioglycolate ester derivatives and these are the mostcommon. The octyl versions are approved (up to amaximum level) for use in rigid food contact and medicalapplications. Sulfur free maleate based tins impartexcellent light stability to clear PVC used in outdoor applications, but require particular lubrication systemsdue to their anti-lubricating effect. Limitations to theiruse include their relatively high cost and unpleasantodour (particularly for the sulfur tins) and their use isnow almost exclusively in PVC-U applications.The performance of organotin mercaptides is not onlybased on the amount of tin metal content, but on theorganotin species, mercaptide ligand chemistry and organic co-stabiliser. The mechanism oforganothiotin stabilisation has been studied extensively.
Mixed Metal Compound:
Liquid mixed metal heat stabilisers are a blend of themetal soaps or salts in combination withorganophosphites and co-stabilisers in a liquidmedium. These materials are used almost exclusively
in PVC-P applications.
Barium Cadamium (Ba Cd)
Barium cadmium based systems (may also include zinc)have been available for many years due to their costeffectiveness in combination with good initial colour andlong-term stability. However, in the European area, theiruse was voluntarily phased out by the PVC industry in2001, due to severe restrictions for environmental andtoxicity reasons concerning cadmium.Cadmium based stabilisers are still used in the USAand Asia Pacific areas, but are coming under increasinghealth and environmental scrutiny.
Barium Zinc (Ba Zn)
Ba Zn based systems now have the majority market sharefor stabilisation of PVC-P in Europe due to extensiveformulation development based on increasing the bariumcontent and the important role of new and existingorganic co-stabilisers. Suppliers include Akros.One issue with liquid stabilisers is the emission ofvolatile components (phenol from the organophosphite,solvent, etc.) during processing and from the end useapplication, e.g., floor and wall coverings. Analytical techniques are now available to detect volatile organiccompounds (VOCs) and so influence stabiliser development. New organophosphite-zinc technology hasalso been introduced to improve this characteristic.
Calcium Zinc (Ca Zn)
Calcium Zinc (Ca Zn)Traditionally less effective as a heat stabiliser, muchwork has been carried out to develop more sophisticatedand higher efficiency heat stabilisers based on Ca Zn.This is based on the ‘concerns’ about barium as a heavymetal although there is no evidence of health or environmental concerns. In addition to further formulation development, complex polyphosphates,new Ca Zn intermediates and new calcium technology have all been promoted.
Potassium Zinc (K Zn)
K Zn stabiliser/kickers are used primarily to activatethe decomposition of blowing agents(azodicarbonamides) for PVC foam. (Analternative approach has been the development of modified azodicarbonamides incorporating an activator system.
Plasticizers:
Plasticisers are added to PVC to achieve flexibility andworkability. The flexibility or softness is determined bythe type of plasticiser and level used in relation to thePVC (phr). They also operate as an internal lubricantbetween the PVC molecules. Based on organic esters,e.g., phthalates, adipates, trimellitates, phosphates, etc.,addition levels can be as low as 20 phr for semi-rigid to100 phr for very flexible applications. The effect on thethermal stability significantly depends on the physical state of the PVC-plasticiser system (260). The relativeamount of crystalline PVC in VC/diethylhexyl phthalate has been identified on the basis of acombination of carbon-13 solution and solid state nuclear magnetic resonance (NMR). The main factors which influence plasticiser choice are determined by the specification requirement of thefinished product and depend on:
• Formulation cost
• Migration/permanence
• Plasticisation effect
• Solvating efficiency of the plasticiser for PVC predicted using Hansen solubility parameters and the Hildebrand solubility parameter
- Volatility
• Plastisol viscosity
• Extraction
The most commonly used plasticisers, together withtheir characteristics, are listed in Table 3.
The fundamentals of plasticiser selection have been listed. The thermodynamics of mixing PVC with phthalate esters have been studied. PVC-plasticiser interactionhas been described using light transmission. The volatile photodegradation products from modelplasticised (dibutyl adipate – DBA) and titaniumdioxide (TiO2 ) pigmented films have been determinedwith CO2 being the main constituent.
The ease with which plasticiser is combined with PVCis a measure of processing characteristics critical inthe dry blend mixing operation of S-PVC or the gelationstage of plastisols. Predictive equations have been developed.
For high temperature performance linked to lowmigration and volatility, trimellitates are used, Plasticisers based on pyromellitic tetraesters have also been proposed, Analytical methods have been developed to dentifymonomeric plasticisers in flexible PVC, PVC-P materials, subjected to ageing under model andclimatic conditions, have been extensively tested.Properties are lost as a result of diffusion controlleddesorption of the plasticiser and a mathematicaldescription of the ageing process has been formulated to predict lifetime. Plasticiser loss on ageing has alsobeen identified in the spectroscopic investigation of roofmembranes in service for a varying number of years, Stain resistance of resilient sheet flooring has beenrelated to the plasticiser, wear layer toughness and thermoplastic nature.
The plasticiser structure, level,extraction resistance, volatility and solubility all have an influence, The phenomenon of plasticiser migration intopolyurethane foam, leading to hardening and cracking in arm and head rest applications, has been investigated and a mechanism proposed.
Phthalate Alternatives
Several alternatives to phthalate esters have beenpromoted, primarily due to the considerable media,
legislative and scientific attention on phthalates. Mostsuggested alternatives already have a niche use inspecific PVC-P applications.
A phenol alkyl sulfonate ester is claimed to replace DEHP. Benzoate esters have already been used for some timeand new blends have been introduced for plastisols.
Di-isononyl-cyclohezane-1,2-dicarboxylic ester(DINCH) has also been developed, Citrate based plasticisers, although much moreexpensive, have particular use in medical and packaging films.
Table : 01 Plasticisers type & it’s applications:
| Plasticisers type | Characteristics | Typical Uses |
| Commodity Phthalate Esters | ||
| C8 Di-2-ethylhexyl phthalate (DEHP)/Dioctyl phthalate (DOP) Di-isooctyl phthalate (DIOP) |
Good fusion rate & viscosity | General Purpose |
| C8 – C10 Di-isononyl phthalate (DINP) Di-isodecyl phthalate (DIDP) |
Slightly less efficient but lower volatility than C8 | Toys & general purpose with good low temperature performance |
| C7 Di-isoheptyl phthalate (DIHP) Dipropylheptyl phthalate (DPHP) |
Good Solvation | Flooring/Coating Plastisol cable |
| C13 Di-isotridecyl phthalate (DTDP) |
High gelation temp. Low extraction, Good migration resistance | High temperature cable insulation |
| Speciality Phthalate Esters | ||
| <C8 Dibutyl phthalate (DBP) Di-isobutyl phthalate (DIBP) Butylbenzyl phthalate (BBP) |
Fast fusing, high volatility, Lower Plasticising efficiency | Flooring |
| Phosphate Esters | ||
| Triaryl phosphates Isobutylated or isopropylated triaryl phosphate esters |
Flame retardant with excellent performance | Electrical cable & general applications, like tents, covers & belts etc. |
| Trimellitate Esters | ||
| Tris-2-ethylhexyl trimellitate/Tri octyl trimellitate (TOTM) Trimellitate ester of mixed semi linear C7 and C9 alcohols (L79TM) Trimellitate ester of mixed C8 and C10 linear alcohols (L810TM) |
Better extraction and migration resistance. Good high temperature, but poorer low temperature performance. |
High specification electrical cable insulation and sheathing. Automotive interior (low fogging). |
| Secondary Plasticisers | ||
| Chlorinated Paraffin | Hydrocarbons chlorinated to level of 40-50%. Have some compatibility constraints and can influence heat stability |
Used in conjunction with primary plasticizers for volume with cost cutting,
Chlorine is help for fire retardancy |
| Epoxidised soybean oil (ESBO) Epoxidised linseed oil (ELO) |
Primary use as a secondary heat stabiliser but also impart flexibility. Compatible only at relatively low addition levels. |
General Use |
| Polymeric Plasticisers | ||
| Polymeric adipates of differing molecular weight. Polyphthalates |
More difficult to process/less compatible but excellent resistance to extraction. High cost. |
Application requiring excellence performance and chemical resistance |
Process Aids
Process aids are added primarily to uPVC to improve fusion characteristics and melt flowduring the processing stage. Addition levels aretypically from 1-6 phr with resultant improvements inoutput and surface finish. Transparency can also beimproved in clear formulations. Chemically, theseproducts are high molecular weight methylmethacrylate and alkyl acrylate copolymers.
The role that acrylic processing aids play in compoundprocessability, end product properties andmanufacturing efficiency has been improved. Acrylic process aids are also played a role in stabilising factor as a co-stabiliser.
Impact Modifiers
Unmodified uPVC has relatively poor impact strengthat, and below, ambient temperatures and the inclusionof an impact modifier significantly improves its
performance. Examples of the different generic typesof impact modifier that can be added to PVCformulations are as follows:
- methyl methacrylate-butadiene-styrene terpolymer(MBS)
• acrylate-polymethacrylate copolymer (acrylic)
• chlorinated polyethylene (CPE)
• acrylonitrile-butadiene-styrene terpolymer (ABS).
Addition levels are typically from 5-12 phr and theirincorporation can influence gelation and melt viscosity.Having limited compatibility with PVC, it is believed that the impact improvement is achieved via theinterruption of the homogeneous morphology into aheterogeneous structure. A disperse phase in the PVC matrix is formed, which creates blockages to crackpropagation and acts as an internal energy absorber.
Acrylic modifiers have good processing characteristicslinked to reduced die swell and good outdoorweathering stability. Their use in window profile andsiding applications are well documented.
CPE impact modifiers contain around 35% of chlorineand are thermoplastic in nature. With a similar meltingpoint to PVC, they initially form a network structure which changes to a particulate structure on processing. They also have good processability and excellentweathering performance; consequently they are also used for window profiles.
MBS impact modifiers are used in a wide range ofapplications, particularly for clear packaging, as therefractive indices are similar between PVC and themodifier. They are not suitable for outdoor applications.Their impact behaviour has been studied in terms ofthe ductile/brittle transition.
ABS terpolymer modifiers also process wellin clear applications giving low crease whitening andexcellent chemical resistance. The heat distortiontemperature of the ABS modified compound can behigher in comparison to MBS modified. They are alsonot suitable for outdoor applications.
Polybutene has also been shown to improve impactresistance and dynamic thermal stability in impactmodified PVC-U formulations.
Heat Distortion Temperature Modification
A major disadvantage of PVC is its relatively lowservice temperature (60-70 °C) and the addition of highlevels of some polymers can improve the heat distortion temperature and Vicat softening point. An example ofsuch a polymer is alphamethyl styrene-acrylonitrile(AMSAN) copolymer. AMSAN-ABS polymer can beused as a combined heat and impact modifier.
Modifiers for Semi-Rigid and Plasticised Applications
CPE modifiers are also suitable for flexible PVCapplications where a high degree of flexibilitycombined with durability is required. Acting as apermanent plasticiser, the liquid plasticiser componentcan be eliminated or substantially reduced.
ABS resins can be added to semi-rigid PVCformulations to improve hot melt strength, elongationand low temperature flexibility.
Lubricants
Lubricants function classically in two ways,(1) externally to reduce friction between the polymermelt and hot metal surfaces during processing, and (2) internally to reduce frictional forces within thepolymer matrix and, in so doing, lower the effective viscosity. External lubricants, by definition, have poorcompatibility with PVC. Internal lubricants are morecompatible and have high polarity. Both types areessential for processing of PVC-U formulations. Inpractice, most lubricants have both external and internalcharacteristics, but vary in the ratio of one to the otherin the way they perform. However, the correct balanceand type of internal to external and addition level arecrucial due to the influence on gelation level, plate-outof incompatible materials in the die and calibrationequipment, and physical properties.
Typical strongly external lubricants are paraffin wax,polyethylene wax, oxidised polyethylene wax andstearic acid. Typical strongly internal lubricants are glycerol esters and fatty acids. Calcium stearatefunctions somewhere between internal and external,depending on addition level. It also acts as an acidscavenger.
Fillers
For most PVC-U and PVC-P applications, fillers areadded primarily to reduce formulation cost, but someare also used to enhance properties and performance.There is a balance between the cost benefits and anyacceptable deterioration in physical properties that could result. The density of the final product willincrease, of course, at higher filler levels. Another important aspect is the influence on processingwith respect to output, plate-out and surface finish.
Calcium Carbonate
Calcium carbonate (also known as chalk) is the mostcommonly used filler for PVC. This material is minedas calcite mineral and ground to a particular particlesize range. It may also be precipitated from solution togive a fine particle size suitable for use in highperformance areas.Particle size is important and, for some applications
requiring good weathering and impact performance(window profile), the ultrafine milled, high whiteness,natural version is normally used.
To ease dispersion,the filler is usually coated with stearic acid. Coatedultrafine and precipitated calcium carbonates areclaimed also to have a positive effect on impact properties in impact modified formulations. The abrasive wear of calcium carbonate, on meltprocessing equipment, is not significant but increases with increasing levels.
Tensile strength properties of PVC-U, filled withprecipitated and ground calcium carbonate, have beeninvestigated.Calcium carbonate nanoparticles arecommerciallyavailable and are claimed to give a cost effective way of increasing impact strength. Their use in impactmodified PVC has improved mechanical properties.
Wood Fillers/Fibres/Flour Composites
The use of wood fillers has expanded considerably,particularly in the USA. In comparison with traditionalproducts, wood filled PVC has superior weatheringresistance and can be used as wood. Finiteelement analysis, of impact durability, has been carriedout to establish the optimum profile wall thickness toresist impact for PVC containing 60% wood fibre.The positive effects of modifiers on physical properties have been identified.
The use of CPE as acompatibiliser, for composites containing 25% and40% wood flour, has been demonstrated with improvedmelt strength and improved surface finish.Accelerated UV weathering performance testing indicated that wood flour are effective chromophoresas they accelerated the degradation of the polymericmatrix. However, there was no deterioration in strengthand stiffness properties.
Other Fillers
Depending on the geographical area, ground marble fillers,some with surface treatment, are used for PVC-U andPVC-P applications.Ground dolomite (calcium magnesium carbonate) and limestone fillers are also used.
Talc has been used in calendered PVC compounds toincrease melt cohesion and modulus of the finishedmaterial.Cellulose fillers, with and without silane coupling agenttreatment, have been investigated at various levels showingan increase in physical properties and viscosity. Silane coupling agents have also been used for interfacialenhancement of PVC-P/silica composites. Calcined kaolin clays can be used in footwear(improved abrasion resistance) and cable (improved insulation properties).
Calcium silicate mineral filler has been proposed as areplacement for calcium carbonate in primary insulation cable compound. Calcined calciumsulfate has been suggested to effectively replace calcium carbonate in cable insulation.
Kaolin has a use in plastisol formulations giving goodrheological control. Calcium carbonate is also used inthis area. An alternative to calcium carbonate, phyllite,has been evaluated. This material, which containsquartz, muscovite and kaolinite, was characterised onthe basis of paste viscosity, gelation and fusionbehaviour, and mechanical properties.
Flame Retardants (FR) and SmokeSuppressants (SS)
PVC-U formulations have low flammability due to thechlorine content. The addition of plasticiser in PVC-Pformulations necessitates the use of flame retardant andsmoke suppressant additives. These additives areknown as functional fillers and a correct balance isnecessary to achieve all the end-use specification requirements. They are predominately used in cable,conveyer belting and roofing membrane formulationsto give resistance to fire initiation and propagation. Itis also important to reduce dripping in a fire situationand that as little smoke as possible is generated.Antimony trioxide has been used extensively, usuallyin combination with phosphate ester plasticisers, giving excellent fire performance and mechanical properties.
The FR mechanism is activated by the formation ofantimony oxychloride which acts as a radical scavengerand flame poison. However, antimony trioxide is asuspected carcinogen and work is ongoing to replaceor reduce the levels used. The use of zinc sulfide hasbeen suggested.
Zinc borate is also an effective SS in combination withphosphate ester plasticiser. The combination ofammonium octamolybdate and zinc stannate is alsobeneficial.
Ethylene copolymers (high molecular weightflexibilisers) have been proposed to replace liquid
plasticiser for low smoke FR PVC cable formulations.
Calcium carbonate and lithium carbonate fillers alsoact as HCl absorbers and the effect on HCl generationduring the combustion of PVC-P has been shown to beeffective, particularly when used in combination.
Pigments
Pigments for PVC must be thermally and light stable,have good dispersibility and be compatible withinthe formulation. Inorganic pigments are the mostcommon type.Titanium dioxide (TiO2) pigments are used to give‘bright’ whiteness and opacity.Specific titanium dioxide grades are used in PVC-Uapplications and contribute to outdoor weathering performance. Their influence on photodegradation, and on the kinetics of weathering have been studied. Reversiblediscolouration effects linked to the photochemicaldegradation of titanium dioxide pigmented PVC, have been shown, after a period of storage of the agedmaterial in the dark. This has been attributed to the formation of particular polyenic sequences, with thescreening effect of the pigment protecting these polyenes against photooxidation, so permitting thesepolyenes to accumulate in the degraded polymer.
Volatile species produced during the UV irradiation ofa white pigmented PVC construction panel have beenmeasured as a function of irradiation time. Rates ofvolatilisation of organic and chloro-organic compoundsdropped sharply with irradiation time. This drop wasattributed to the crosslinking of the irradiated surfacewhich became chlorine deficient in TiO2 pigment.
Pinking of PVC profiles, in mild climatic conditions,has been observed in service. This has been explainedby the formation of polyene sequences not completelyphotooxidatively bleached under conditions of oxygenstarvation. One predictive test has been designed and validated. Other predictive tests have alsobeen suggested.Titanium dioxides for use in PVC plastisols must satisfythe need for good wettability and dispersion within theliquid medium. Paste storage stability is also important.
The effects of different surface treatments on TiO2 on these parameters have been studied, with the conclusionthat the amount and type of alumina treatment is significant.
Aluminosilicates, as pigmentextenders to partially replace TiO2 in plastisols, havebeen investigated, with whiteness retention andreduction in surface gloss.
Lead chromate pigments, traditionally used for pipeand cable applications, are linked with the phase outof lead based stabilisers. Special pigment systems basedon organic or organic/inorganic combinations areavailable.
Biocides
PVC-P materials, such as flooring and roofing material,can be prone to microbiological attack in humid ordamp conditions. This is due to the fungi using theplasticiser at the surface of the article as a food source.This can lead to partial discolouration (pink colour orblack specks) which can further cause a tacky surfacewhere dirt can accumulate. Unpleasant odours may alsobe a consequence.
Biocides function by becoming active on the surfaceof the material to destroy the fungi. Plasticiser transferto the surface is limited by the process of diffusion ofthe plasticiser within the material, the fungus also actingas a leaching solvent.
Blowing Agents
Solid blowing agents are materials which decomposeto release gases at particular temperatures matchingthe appropriate melt viscosity necessary to retain thefoam structure. There are two main types:(1) azodicarbonamide, which is exothermic in natureand decomposes to give primarily nitrogen gas and(2) sodium bicarbonate, which is endothermic and soabsorbs heat as it releases carbon dioxide gas. Variousgrades are available to suit particular applications andboth have well controlled particle size distribution. Theazo compounds can be specially formulated, to include an activation agent which catalyses the decompositionand so increases the total gas evolved. Granular formsimprove handling.
PVC-U foams incorporate sodium bicarbonate and theresults are available of an investigation into controllingthe rate of gas evolution and heat of decomposition.The manufacture of cellular PVC/wood composites hasbeen studied. The properties achieved, as foam densitywas reduced, were examined showing suitability formany wood replacement applications.
Antioxidants and Light Stabilisers
Primary antioxidants, such as hindered phenols, operateas effective radical scavengers to protect the PVCmaterial during processing and in use (preventingphotodegradation). Phosphites and thiosynergists arealso used as secondary antioxidants to extend theefficiency of the primary antioxidant by reduction ofoxidation intermediates. These can be added at verylow levels at the polymerisation stage and incombination with the stabilising package at the blending stage.
Light stabilisers also prevent photodegradation. UVabsorbers such as hydroxybenzophenone or
hydroxyphenyl triazole types, operate by absorbing anddissipating UV radiation prior to potential degradationof the polymer. No permanent chemical change occurs,so activity is retained. Hindered amine light stabilisers(HALS) are also used but their activity is notparticularly well understood.
Cyanoacrylates have been proposed as a new range ofUV absorbers for PVC and other plastics.
Bonding Agents
PVC plastisol, spread on to industrial fabrics based onpolyester or polyamide fibres, requires the addition ofa chemical bonding agent to improve the interfacialadhesion. Based on polyisocyanurates, dispersed in aplasticiser, this functions by reacting with the polargroups in the synthetic fibre to give strong chemicalbonding at the interface of the fabric and PVC. Thesematerials are highly reactive and the plastisolformulation has to take this into account in relation toplastisol viscosity build-up