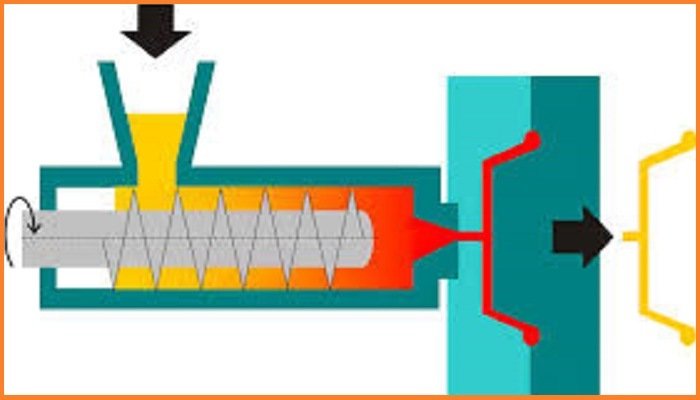
Low-density polyethylene
Low-density polyethylene (LDPE) is a thermoplastic made from the monomer ethylene. It was the first grade of polyethylene.
Properties
LDPE is defined by a density range of 0.910–0.940 g/cm3. It is not reactive at room temperatures, except by strong oxidizing agents, and some solvents cause swelling. It can withstand temperatures of 80 °C continuously and 95 °C for a short time. Made in translucent or opaque variations, it is quite flexible and tough.
LDPE has more branching (on about 2% of the carbon atoms) than HDPE, so its inter molecular forces (instantaneous-dipole induced-dipole attraction) are weaker, its tensile strength is lower, and its resilience is higher. Also, because its molecules are less tightly packed and less crystalline due to the side branches, its density is lower.
Chemical resistance
- Excellent resistance (no attack / no chemical reaction) to dilute and concentrated acids, alcohols, bases and esters.
- Good resistance (minor attack / very low chemical reactivity) to aldehydes, ketones and vegetable oils.
- Limited resistance (moderate attack / significant chemical reaction, suitable for short-term use only) to aliphatic and aromatic hydrocarbons, mineral oils, and oxidizing agents.
- Poor resistance, and not recommended for use with halogenated hydrocarbons.
Applications
LDPE is widely used for manufacturing various containers, dispensing bottles, wash bottles, tubing, plastic bags for computer components, and various molded laboratory equipment. Its most common use is in plastic bags. Other products made from it include:
- Trays and general purpose containers
- Corrosion-resistant work surfaces
- Parts that need to be weldable and machinable
- Parts that require flexibility, for which it serves very well
- Very soft and pliable parts such as snap-on lids
- Six pack rings
- Packaging for computer hardware, such as hard disk drives, screen cards, and optical disc drives
- Playground slidess
- Plastic wraps